The drug was developed by the company Revolution Medicines, and is still under regulatory review. The approval given now does not make it an approved drug for wide use, but allows it to be given in a framework known as expanded access, or compassionate use, to patients who meet the criteria and for whom there is no sufficient therapeutic alternative. According to the company’s statement, the referral to the route will be made through the attending physician only, and the company stated that it will provide the drug free of charge to patients participating in the program.
The data that aroused great interest in the oncology community comes from a phase 3 study called Resolute 302, in which the drug was tested in patients with metastatic pancreatic cancer that had already been treated. According to the company’s announcement, the median overall survival among patients who received draxonersib was 13.2 months, compared to 6.7 months among patients who received chemotherapy according to the doctor’s choice. The study met its main objectives, including overall survival and survival without disease progression, and its findings are expected to be presented at the annual meeting of the American Society of Clinical Oncology.
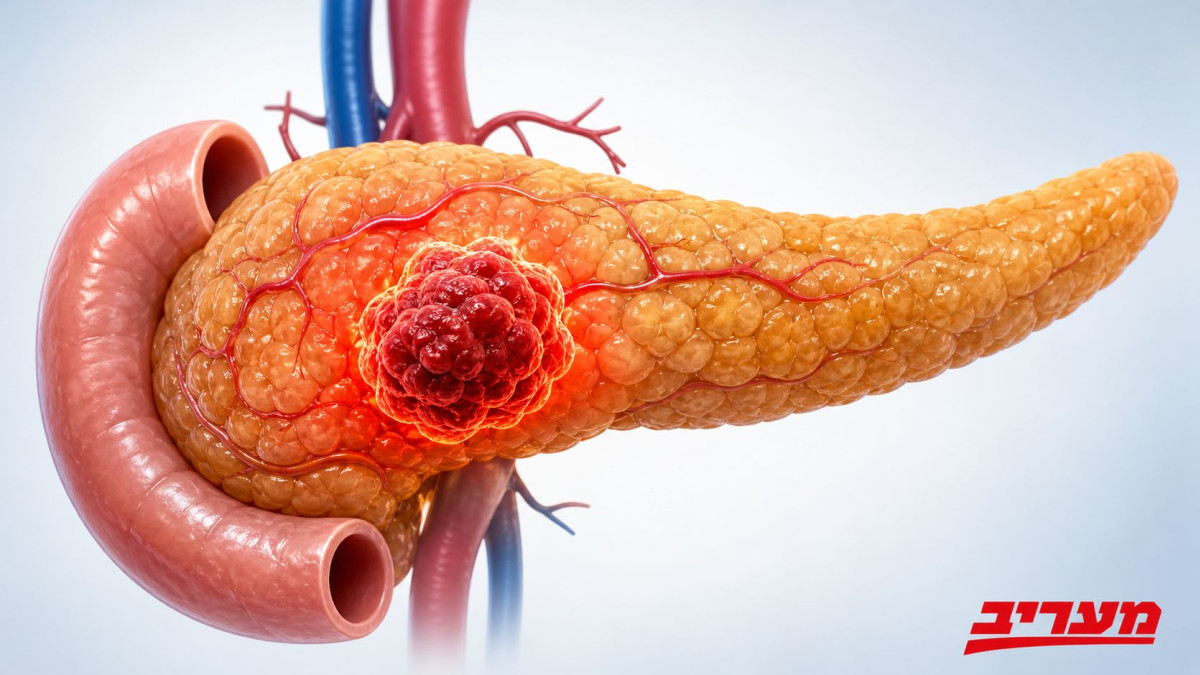
The action of the drug focuses on one of the important growth mechanisms of pancreatic tumors. Derxanersib is an inhibitor of Ras proteins in their active state. The Ras protein family acts in the cell as a kind of biological switch: when the switch is turned on, it transmits conserved signals to the cell to divide, survive and grow. In normal cells this activation is controlled, but in a variety of cancerous tumors, and especially in pancreatic cancer, mutations in this pathway cause the signal to remain active for a long time. This creates a continuous drive for cancer cell proliferation, tumor progression and resistance to treatments.
For years, Ras proteins were considered a particularly difficult target for drug development. Their structure has made stable drug binding difficult, and they operate within a complex network of cellular signaling pathways. In recent years, drugs aimed at giving certain types of Ras mutations have begun to appear, but pancreatic cancer has posed an especially big challenge: in more than 90% of cases of adenocarcinoma of the pancreatic ducts, there are mutations in the K-Ras gene, which are one of the main forces driving the disease.
The uniqueness of Draxonresib is in trying to inhibit the Ras proteins when they are in the active state, and not focus only on one rare mutation. In this way, it is intended to block the wider cancer signaling pathway, which is behind the tumor cell culture. Simply put, the drug tries to take the leg off the biological gas of the cancer cell. If the signal that tells the cells to divide is weakened, the tumor may progress more slowly, and the patient may gain more survival time.
Despite the encouraging data, it is important to emphasize that draxonarsib is still defined as an experimental drug. This means that the full information on its effectiveness and safety is still being tested, and access to it at this stage is limited to only certain patients in the United States. The expanded access route of the Food and Drug Administration is intended for situations where it is a serious or life-threatening illness, when there is no adequate alternative treatment, and when there is medical justification to allow early access to the drug even before full approval.
Pancreatic cancer is considered one of the most difficult types of cancer to diagnose and treat. The pancreas is located deep in the abdominal cavity, behind the stomach, so tumors in it may develop for a long time without causing obvious symptoms. When signs appear such as unexplained weight loss, stomach or back pain, jaundice, loss of appetite, weakness or the onset of new diabetes, the disease may already be in an advanced stage. This is one of the reasons why a large proportion of patients are diagnosed when the tumor is not resectable or has already metastasized.
The data in the United States illustrate the seriousness of the disease: according to the American Cancer Society, in 2026, approximately 67,530 people are expected to be diagnosed with pancreatic cancer, and approximately 52,740 are expected to die from the disease. The relative survival rate for 5 years in all stages of the disease is only 13%. When the disease is detected at a local stage, the 5-year survival rate is higher and stands at 44%, but in metastatic disease it drops to only about 3%.
Pancreatic cancer treatment depends on the stage of the disease, the general condition of the patient and the characteristics of the tumor. When surgery is possible, removing the tumor is the only option that may lead to recovery, but only a small proportion of patients are suitable for surgery at the time of diagnosis. Many are treated with chemotherapy, sometimes in aggressive combinations, and in some cases combine radiation, supportive treatments or tailored treatments based on genetic testing of the tumor. In recent years, the trend of adjusting treatment according to mutations has been strengthening, but in pancreatic cancer the progress has been slow compared to other types of cancer.
Precisely for this reason, the data on draxonarsiv arouse unusual interest. Doubling the median survival in a previously treated metastatic disease is a significant figure, even if it is an addition of months and not a cure. For patients with advanced disease, any extension of life of reasonable quality can be of great importance. However, only full publication of the study data, including the rate of side effects, quality of life, duration of response and the characteristics of the patients who particularly benefited from the treatment, will make it possible to assess the exact place of the drug in the therapeutic arsenal.
The Food and Drug Administration emphasized that the move reflects recognition of the urgent need for new treatments for a life-threatening disease. The company’s request for extended access was submitted on April 28, and approval was given on April 30, that is, within just two days. This is an unusual speed relative to regulatory procedures, and it reflects the balance that the regulator tries to maintain between scientific caution and the need to allow therapeutic hope for patients who have exhausted existing options.
At this stage, this is not immediate news for patients in Israel, as long as the drug has not been approved for marketing and has not been registered in the local routes. However, if the approval process in the United States moves forward later this year, and if the full data confirm the initial findings, Draxonorsib may become one of the first drugs that succeed in actually changing the course of the disease in a particularly difficult group of metastatic pancreatic cancer patients after previous treatment.